
Sara System
Inventory Management



Information
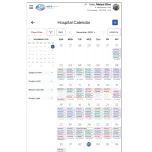
The SARA System® is an advanced hospital management solution for surgical centers and central sterile services departments (CSSD), featuring a patented, innovative traceability technology. Its unique capability allows it to verify the completeness of surgical trays even when they are closed and sealed.
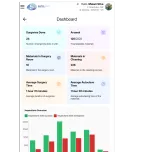
The SARA System® elevates surgical asset management using an all-in-one platform of hardware, software, and AI. It provides real-time digital traceability, automatic kit verification, and guaranteed regulatory compliance. Our system reduces operational costs by minimizing waste and human error while optimizing inspection times. Integrated with the surgical calendar, it delivers a constantly updated inventory and actionable insights to drive efficiency, enhance patient safety, and improve hospital profitability.
The SARA System® provides a comprehensive and integrated solution for optimized surgical center management. It incorporates an intelligent surgical schedule, Sara TV for interactive communication between the surgical center and the materials and sterilization department, and rigorous control over respiratory kit assembly. Furthermore, the system features complete autoclave management and an internal process methodology that encompasses the entire ecosystem of surgical instruments. All these functionalities are enhanced by proprietary software with integrated artificial intelligence, which delivers detailed reports and KPIs for strategic and efficient management.
