
Drain-TG™ - JSR Medical
Abdominal Surgery

Information
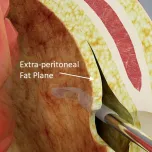
Drain-TG™ enables anastomotic leakage (AL) monitoring and immediate treatment of AL without surgical intervention by preventing drain migration through an extra-peritoneal tunneling drain insertion.
[FDA Registered]
- Indication: Guide, Surgical, Instrument
- FDA Device Listing #: D480270
- FDA Product Code: FZX
- FDA Device Class: Class I
[Development Background]
Anastomotic leakage after surgical procedures is a major risk factor that can lead to complications such as sepsis.
Therefore, drain tubes have to be placed before anastomotic leakage to prevent complications.
The conventional way of inserting the drain tube is using laparoscopic instruments, and there is a problem that it is not fixed and may migrate.
JSR Medical has developed a new drainage system that utilizes the extra-peritoneal tunneling drain insertion to guide the drain tube to be inserted and secured at the anastomotic site, thereby minimizing complications.
[Indication for Use]
- A single-use device for immediate diagnosis and treatment of anastomotic leakage at the anastomotic site.
- Use the extra-peritoneal tunneling method to fix the drain tube at the anastomotic site.
