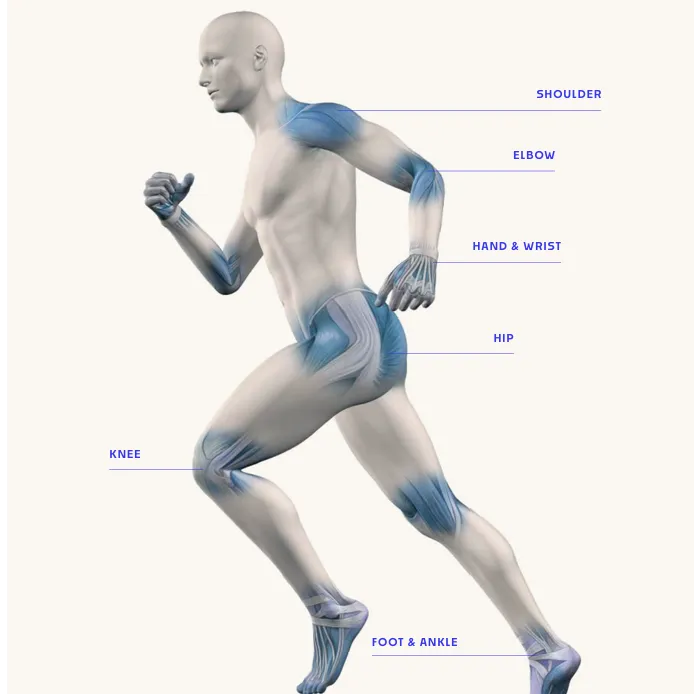
Auxillock Sports Medicine Products
Internal, External Fixation, Support Immobilisers

Information
SPORTS MEDICINE CATALOGUE
The content of this catalogue is intended for healthcare professionals only. All product
information, illustrations, descriptions, and specifications provided here are for gen
eral informational purposes and are subject to change without notice. Auxein Medical
Pvt. Ltd. reserves the right to improve or discontinue products at any time without prior
notification.
This document does not constitute medical advice. Surgeons and medical practitioners
must exercise their own professional judgment when using any medical devices or in
struments, including those provided by Auxein.
INTENDED USE
The implants and instruments in this catalogue are intended for use by qualified ortho
pedic surgeons trained in arthroscopy and reconstructive procedures. Improper selec
tion, misuse, or lack of experience may result in complications.
REGULATORY COMPLIANCE
US FDA 510(k)
EU MDR
CE Mark (Class IIb)
MDSAP (Medical Device Single Audit Program)
ISO 13485:2016 Quality Management System
CDSCO – Indian Medical Device Rules, 2017
INTELLECTUAL PROPERTY
All trademarks, trade names, brand names, product names, and logos mentioned in
this catalogue are the property of Auxein Medical Pvt. Ltd. Unauthorized use or repro
duction is strictly prohibited
