Radiopharmaceuticals for Diagnostics an Therapy
Pharmaceutical Products, Prescriptions
Information
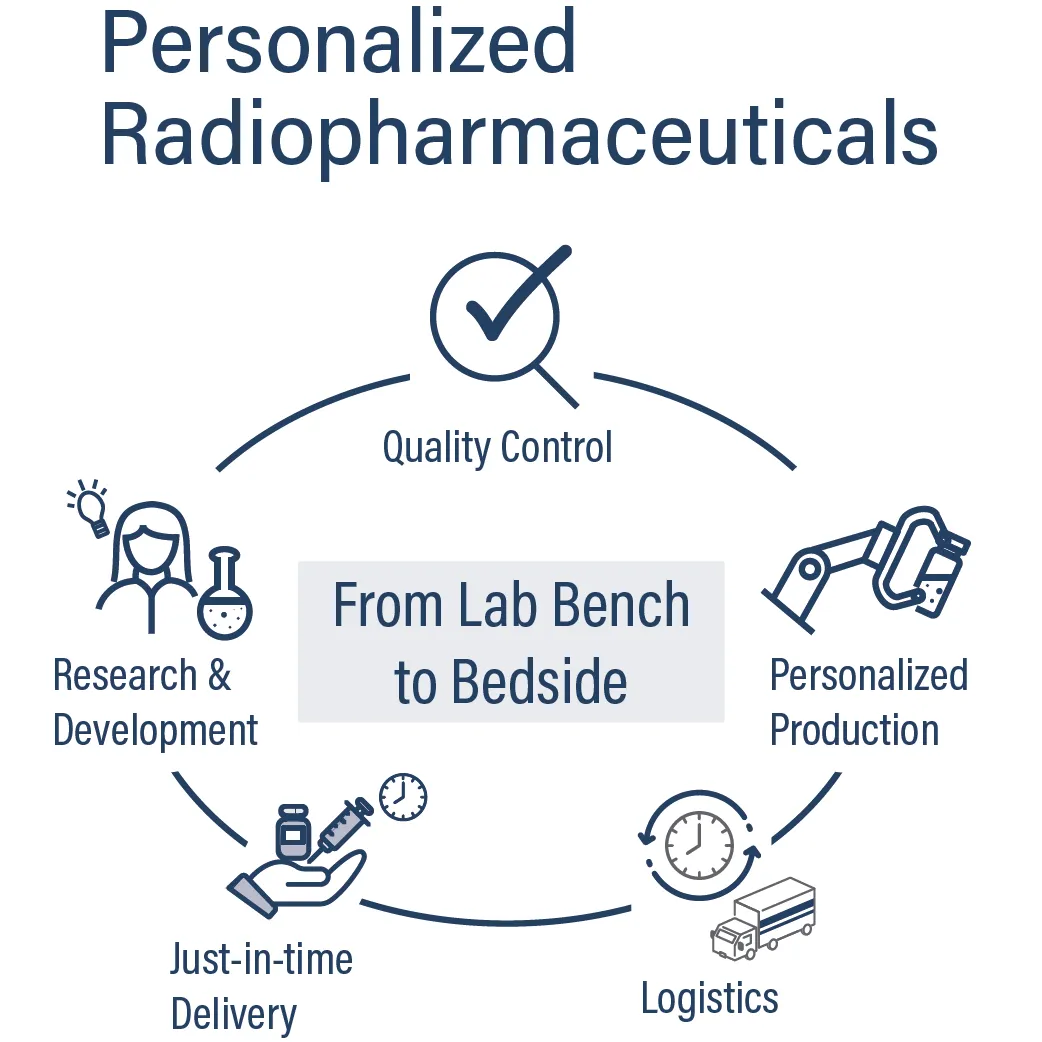
We produce radiopharmaceuticals in six highly specialized GMP cleanroom laboratories with a total area of 400 m² at our site in Seibersdorf, Austria, one of the leading radiopharmaceutical production sites in Europe.
As a European radiopharmaceutical CDMO, we support both clinical development programs and commercial manufacturing, offering pharmaceutical companies a reliable production partner with short decision-making processes and fast time-to-market implementation.
Product & Service Portfolio
Our radiopharmaceutical CDMO services in Europe:
• Radiolabeling of small molecules, peptides, and antibodies
• Formulation and stabilization of radiopharmaceutical active ingredients
• Aseptic filling under GMP conditions
• Packaging and release processes in accordance with European and international regulatory requirements
Our products:
• Investigational medicinal products (Phase I–III) for early and late development phases
• Commercial radiopharmaceuticals for the European and international markets
Our current focus is on diagnostic and therapeutic radiopharmaceuticals (theranostics) using the radionuclides fluorine-18 (F-18) for PET diagnostics, lutetium-177 (Lu-177) for targeted radionuclide therapy, and actinium-225 (Ac-225) for alpha-therapeutic applications in oncology.
